A piece of “Ice No. 7” is enough to freeze all the oceans on earth? What exactly is “Ice No. 7”

Cryptozoology: We’re still finding prehistoric survivors Ever since the news of the ability to wear short sleeves in the Arctic was reported, everyone has started to worry about the situation of the ocean, thinking that under this trend, it will not take long for it to invade the land with great fanfare and completely devour the homes built by people. At this time, some people opened their brains and pointed out that it is easy to solve this problem, just find a way to freeze all the oceans of the earth
The earth may become a “hockey puck” by then
You may think that this idea is too outrageous, and science fiction films dare not act like this. But in fact, it is possible to do it, a piece of “No. 7 ice” is enough. So, what is this “No. 7 ice”? Could it freeze all of Earth’s oceans?

Is there such magical ice on the earth?
Ice has many phases
When it comes to No. 7 ice, many people must be full of question marks. How can ice made of water be divided into “three, six, and nine grades” by humans? We have never heard this name in our daily life. Scientists have been classifying ice for a long time. The reason for classification is that from a microscopic perspective, there are indeed great differences between various types of ice.
If you just observe from the surface, it is difficult to see the differences between various types of ice. It is necessary to analyze the crystal structure of ice in the microcosm.
According to this standard, the ice that we often see in our daily life belongs to No. 1 ice. Because of its internal structure, people also call it hexagonal ice.
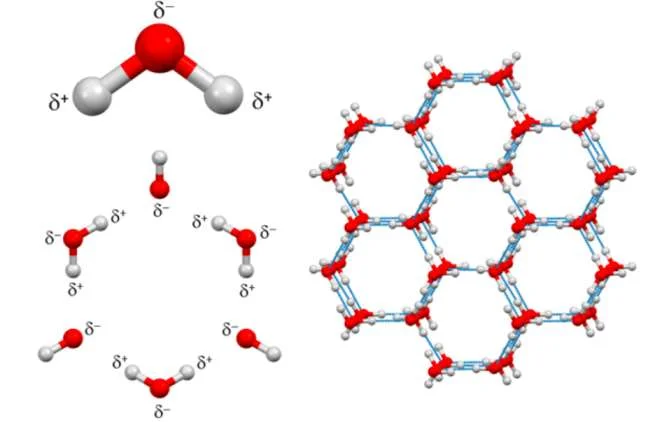
Schematic diagram of the molecular structure of No. 1 ice
Of course, at first, people thought that the ice in nature should be “mass-produced”, in short, they are all the same. But later everyone realized that when the temperature and pressure change, the hydrogen bonds between water molecules will show different grid structures and strengths, thus presenting many complex ice phases, and each ice phase Ice has its characteristics.
According to the current research, humans have discovered more than a dozen ice crystal structures, most of which were obtained in the laboratory. Because in the natural environment, it is difficult to make the pressure and temperature that meet people’s requirements appear at the same time. Among them, only the hexagonal phase ice Ih and the cubic phase ice Ic can form and exist naturally in nature.
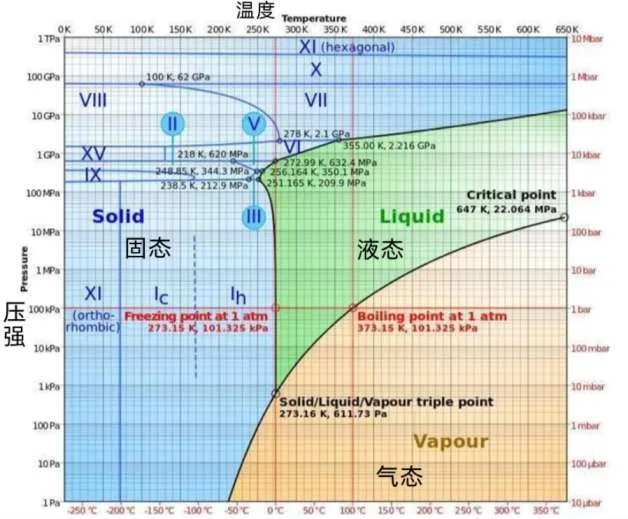
Schematic diagram of the three-state change conditions of water
In this way, people began to explore the ice phase of ice since 1900. At that time, people obtained No. 2 ice with hydrogen order through simple pressurization. However, due to poor observation equipment, the structure of Ice No. 2 was not analyzed by single crystal X-ray diffraction until 1964.
Later, as the experimental equipment became more and more perfect, and the observation equipment became more and more advanced, people discovered more ice phases, including the No. 7 ice we are going to talk about today, so how was No. 7 ice discovered? Where is it sacred?

- Is No. 7 ice different from ordinary ice?
The magical “No. 7 Ice”
First of all, one thing needs to be explained, that is, when scientists explore ice phases, they do use numbers to name their order, but they generally add Roman numerals after the ice. For example, ice No. 7 will be expressed as ice VII. Therefore, whether it is ice No. 7 or ice VII, they all refer to the same thing in essence. Read More:- Xishuangbanna has 10 locations, 8 of which are free to visit with a ticket
WhatsApp Bios: Here, we will show you 500+ Best Love, Life, sadness, attitude, and motivation WhatsApp bio Caption ideas in English for boys and girls. Express your feelings with the help of these WhatsApp Bios quotes. These quotes tell us a lot about life. You read these quotes(WhatsApp Bio Caption), and you will also become crazy about them. If you like these Whatsapp captions, definitely put them in your Whatsapp bio. You can also put these beautiful Bio quotes in your Facebook and Instagram bios. Don’t forget to share these Whatsapp statuses With your friend.
According to the data, Klotz, the discoverer of No. 7 ice, found in the laboratory that by decompressing ice VI in an environment below 95K, high-pressure phase ice VII can be obtained. And after that, some researchers said that No. 7 ice can exist stably at a pressure of 22kbar. As for the internal structure of ice 7, it is composed of two cubic ice lattices that are mutually structured, and there are no connecting hydrogen bonds between the sublattices.

Schematic diagram of the molecular structure of No. 7 ice
If you compare it with the common No. 1 ice, you will find that the density of No. 7 ice is very high, about 1.5 times that of No. 1 ice. Looking at it this way, Ice No. 7 is far more “solid” than we imagined. In addition, No. 7 ice has a higher freezing point than No. 1 ice.
As we all know, generally speaking, water will freeze when it is below 0 degrees Celsius. However, No. 7 ice is representative of the slanted front. It can freeze at 4.9 degrees Celsius. From the temperature alone, it seems to be easy to appear in daily life.

Normally, the temperature at which water freezes is 0 degrees
But only the temperature is not enough. As we just said in the previous article, the change in the ice phase is affected by the joint action of pressure and temperature. Although the freezing temperature of No. 7 ice is relatively high, its requirements for pressure are also ridiculously high.
From the research data, it takes a pressure of 3 billion Pa to freeze a piece of No. 7 ice, and our standard atmospheric pressure is only 101.325 kPa. Even if we enter the deepest Mariana Trench on the surface, it is still far from the figure of 3 billion Pa.
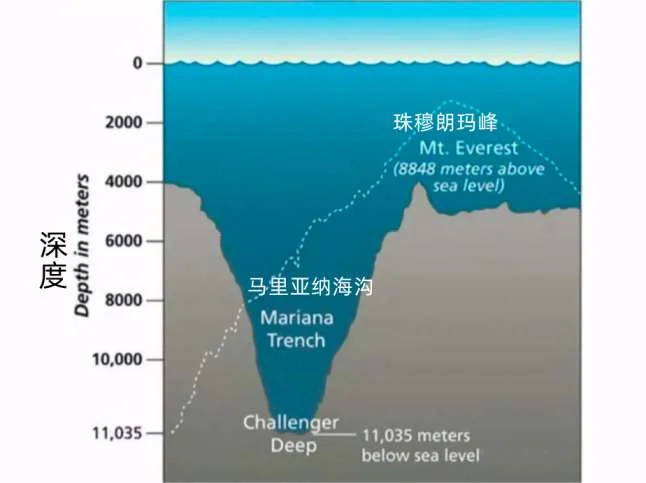
The pressure at the deepest part of the trench can reach 110 MPa
Of course, the world is full of wonders. Scientists originally thought that No. 7 ice would definitely not be found in the natural environment, but unexpectedly, in 2018, an American research team accidentally discovered a diamond. Although diamonds are precious, there is nothing unusual about them in essence. The problem is that rare No. 7 ice appears in diamonds.
It turns out that this diamond has unexpectedly become a laboratory where No. 7 ice freezes. You must know that it can not only meet the needs of No. 7 ice in terms of pressure but also maintain a certain temperature by locking it in the body. below the limit. In this case, a rather mini Ice 7 was born.

Diffraction patterns of ice 7 in diamonds
Scientists were thrilled to find out, but some were concerned. Because in many imaginative descriptions, the existence of No. 7 ice is not good for human beings. On the contrary, it may be used by criminals as a weapon to destroy the earth. Read More:- The Taoranting Ice and Snow Carnival welcomes guests, and the cute little penguin appears again
Could it be that just a piece of No. 7 ice can freeze the earth’s oceans directly?

Humans may not need to learn to swim by then.
Could Ice No. 7 Freeze the Oceans?
The statement that No. 7 ice can freeze the ocean cannot be regarded as “nonsense”, because the characteristics of No. 7 ice are indeed more in line with this assumption.
According to the research of scientists, if Ice No. 7 formed under natural conditions, and the air pressure and temperature at that time met its needs to continue to freeze, then this guy would spread crazily from that “point”. In just one hour, even the nearby 1,600 kilometers can be completely frozen.

- Ice No. 7 under computer simulation
That’s why some people think that if you give people a piece of No. 7 ice, under the right conditions and time, it can indeed freeze the entire ocean. However, through the above introduction, everyone also understands that temperature alone is not enough, and the surface of the earth does not have the pressure required for the formation of No. 7 ice.
In this case, even if someone dropped a complete piece of No. 7 ice into the sea, the expected rapid freezing of the ocean would not occur. A more likely scenario is that Ice 7 melted by the time it entered it.

It’s over before it even starts…
Of course, the fact that such an environment does not exist on Earth does not mean that it does not exist on other planets. According to scientists’ research, exoplanets more than 20 light-years away from Earth may have the potential to breed No. 7 ice. After all, the pressure and temperature on this planet can satisfy No. 7 ice, and more importantly, it seems that there is no land on its surface, but it is surrounded by oceans.
In short, in theory, No. 7 ice does have the ability to freeze the ocean. But if it wants to do this, there are too many preconditions, and the earth obviously cannot satisfy them. Therefore, it is impossible to freeze the ocean of the earth with only a piece of ice.

“Want to freeze me? Practice again!”
It is worth mentioning that, in addition to the No. 7 ice, which has the unique feature of “quick freezing”, which is often cited by people, there is also a magical No. 18 ice, which has been on the hot list of discussions since it was discovered.
Superionic Ice Phase-Ice No. 18
If it is shocking that No. 7 ice can condense at room temperature, then No. 18 ice will subvert our cognition again. As early as 1988, physicists obsessed with the study of ice phases gave a prediction about “high-temperature icing”.
Although it seems incredible, he believes that in an extreme environment where high temperature and pressure exist simultaneously, oxygen ions can be locked in a solid cubic lattice. As for hydrogen ions, they can move freely in them.
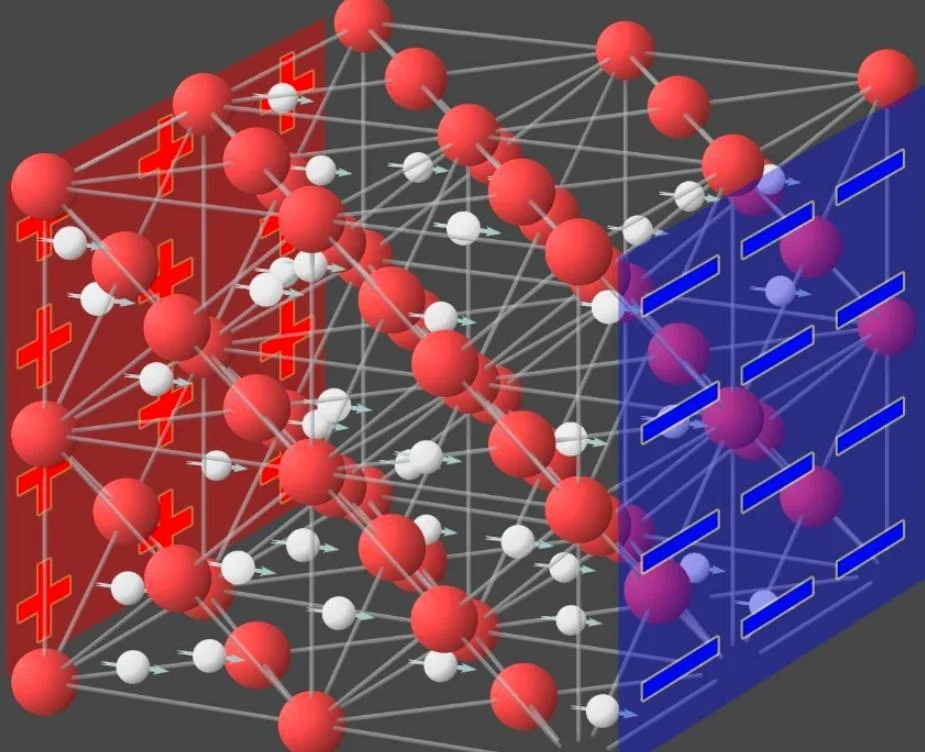
Schematic diagram of the molecular structure of ice 18
In this case, the ice crystal structure would be more stable and have a higher melting point. Thirty years later, this prophecy was finally confirmed.
According to reports, in 2018, LLNL physicist Millot and others found the first clue of the existence of superionic ice. They compressed liquid water so that it turned into solid ice in nanoseconds. The conductivity has increased hundreds of times in a short period of time, which means that the water ice has become a “superionic” state.

- What would superionic ice look like?
Similar to the physicist’s prediction, when the pressure is between 249 and 278GPa and the temperature is between 2000 and 3000K, ice No. 18 can appear smoothly. From this situation, this piece of ice has achieved “rebirth from the ashes”.
| Google News | Follow | Click Here |
| Telegram | Follow | Click Here |
| Quora | Follow | Click Here |
| Follow | Click Here | |
| Follow | Click Here | |
| Dailyhunt | Follow | Click Here |








